PDD
AVIAN BORNAVIRUS INFECTION AND PROVENTRICULAR DILATATION DISEASE
Proventricular dilatation disease (PDD) is a progressive and often fatal disease of birds that primarily affects the nervous system and gastrointestinal tract. Early on, this poorly understood disease appeared to affect primarily macaws and was referred to as Macaw Wasting Disease. It is now known to occur in more than 50 bird species, including cockatoos, cockatiels, parakeets, Amazon parrots, conures, eclectus parrots, African grey parrots, and lovebirds. Cases have also been reported in non-psittacine species such as canaries, finches, honeycreepers, waterfowl, toucans, and birds of prey. This disease seems to affect adult birds more commonly than juveniles.
In 2008, avian bornavirus was experimentally confirmed as the cause of PDD. Although many aspects of this disease in companion birds are still being studied, research suggests that approximately 15% to as many as 33% of companion birds may carry the virus.
TRANSMISSION
Transmission of avian bornavirus is not fully understood, but it is believed to occur primarily through direct contact with infected birds or exposure to their droppings, feather dust, or bodily secretions. Both horizontal transmission (between birds) and possible vertical transmission (from parent to offspring) have been suggested. Some birds may carry and shed the virus without showing clinical signs, making control challenging, particularly in multi-bird environments. The incubation period for avian bornavirus infection can be as brief as days but may also be much, much longer, possibly decades, in some birds. Infected birds are thought to shed the virus for life; however, there is a report of a captive flock of avian bornavirus infected cockatiels spontaneously eliminating the virus, becoming PCR negative for the virus.
CLINICAL SIGNS IN BIRDS
The pathology of avian bornavirus involves a range of clinical signs that can affect both the gastrointestinal and central nervous systems. Because of this, clinical signs can vary wildly between birds. Not all birds display classic gastrointestinal signs. Some birds develop neurological deficits which may occur with or without digestive issues.
When the virus causes inflammation of nerves that control the muscles of the proventriculus (the glandular portion of the stomach) and the gizzard (the muscular portion of the stomach), there is slow or absent movement of food through the digestive tract and a lack of ability to properly digest and absorb nutrients. Birds often lose weight despite a good appetite. Other signs include emaciation, regurgitation, and the presence of undigested food in the feces (see Fig. 1). Additional findings may include abdominal enlargement, lethargy, weakness, diarrhea, reduced fecal output, or increased fluid content in the droppings.

When the virus causes inflammation of the brain or central nervous system, the bird will show neurological signs such as poor balance, uncoordinated walking, blindness, tremors, weakness, head tilt, and seizures.
The progression of the disease can vary significantly between birds as well. Some birds may show only subtle signs early in the disease, while others may deteriorate rapidly.
DIAGNOSIS
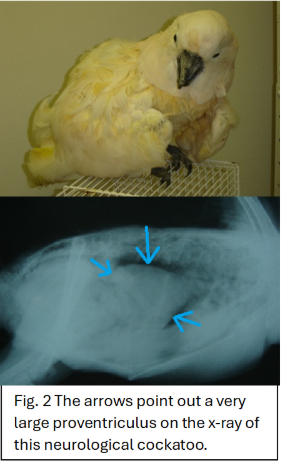
Diagnosis of PDD can be challenging and typically involves a combination of clinical signs, imaging, and laboratory testing. Radiographs or contrast studies may reveal an enlarged proventriculus (see Fig. 2). Polymerase chain reaction (PCR) testing can detect avian bornavirus in blood, cloacal, or choanal swabs, although intermittent shedding can lead to false negatives. Definitive diagnosis is often made through biopsy of affected tissues (such as the crop or proventriculus) demonstrating characteristic lymphoplasmacytic inflammation of the nerves. In some cases, diagnosis is confirmed post-mortem.
TREATMENT OPTIONS
At present, there is no known treatment that eliminates the virus or reduces viral shedding. Instead, treatment options focus on supportive care and management of clinical signs. Nonsteroidal anti-inflammatory drugs (such as celecoxib or meloxicam) are commonly used to reduce inflammation, and some birds may show temporary improvement. Supportive care may include easily digestible diets, fluid therapy, and nutritional support. Prokinetic medications and other therapies may be used depending on clinical signs. Long-term prognosis is generally guarded to poor, although some birds may live for extended periods with supportive care.
WHAT TO DO AT HOME
Effective cleaning and disinfection are important in managing the spread of avian bornavirus. Organic material should be removed prior to disinfection, and the virus is susceptible to most detergents, disinfectants, and sunlight/ultraviolet light. Good hygiene practices, minimizing dust and feather debris, and avoiding overcrowding are important preventive measures. Quarantine of new or exposed birds and routine health monitoring are strongly recommended.
ZOONOTIC CONCERNS
Currently, avian bornavirus is not considered a zoonotic risk, and there is no evidence that it can be transmitted from birds to humans. However, as with all animal diseases, good hygiene practices—including handwashing after handling birds or cleaning enclosures—are recommended to reduce the risk of exposure to other potential pathogens.
IN SUMMARY
When avian bornavirus infection is diagnosed or suspected in a multi-bird household, isolate infected birds whenever possible. Wash your hands between birds and always care for birds that have tested negative for bornavirus infection before handling bornavirus-positive birds. Although avian bornavirus infection is common in clinically healthy birds, the development of clinical disease (i.e., PDD) appears to be more rare.
Leticia Materi PhD, DVM
Calgary Avian & Exotic Pet Clinic


